Unveiling the Chemistry of 2,2-Dimethylcyanoacetate Methyl Ester
2023-10-17
Introduction
In the realm of organic chemistry, a diverse array of compounds exists, each with its unique properties and applications. One such compound is 2,2-Dimethylcyanoacetate Methyl Ester, a mouthful to pronounce but fascinating to explore. In this blog, we will delve into the intricacies of this compound, its chemical structure, synthesis, and potential applications in the world of chemistry.
Understanding the Chemical Structure
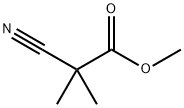
2,2-Dimethylcyanoacetate Methyl Ester is a compound with a complex but intriguing chemical structure. Let's break it down:
1. 2,2-Dimethyl: This part of the name indicates the presence of two methyl (CH₃) groups on the carbon atoms. These methyl groups are attached to the carbon atom at the second position in the molecule.
2. Cyanoacetate: The "cyano" group refers to the presence of a cyano group (CN), which consists of a carbon atom triple-bonded to a nitrogen atom. The term "acetate" signifies the presence of an acetate group (CH₃COO), which contains two carbon atoms bonded together.
3. Methyl Ester: The compound also includes a methyl ester group (COOCH₃), which consists of a carbonyl group (C=O) bonded to an oxygen atom (O) and further bonded to a methyl group (CH₃).
Synthesis and Preparation
2,2-Dimethylcyanoacetate Methyl Ester is typically synthesized through chemical reactions involving various starting materials and reagents. One common method of synthesis involves the reaction between cyanoacetic acid and diazomethane. During this reaction, diazomethane replaces one of the hydrogen atoms in the cyanoacetic acid molecule, resulting in the formation of 2,2-Dimethylcyanoacetate Methyl Ester.
Applications in Chemistry
This compound finds applications in various areas of chemistry, and here are a few notable ones:
1. Organic Synthesis: 2,2-Dimethylcyanoacetate Methyl Ester is frequently used in organic synthesis as a building block for the preparation of more complex molecules. Its versatile chemical structure makes it a valuable starting material.
2. Medicinal Chemistry: Some derivatives of this compound have been explored for potential pharmaceutical applications. Researchers investigate its properties to design new drugs and understand its potential biological activities.
3. Material Science: Certain derivatives of this compound can be used in the development of materials with specific properties, such as polymers or coatings.
4. Chemical Research: Chemists and researchers use this compound to study reaction mechanisms, chemical kinetics, and various aspects of organic chemistry.
Conclusion
While 2,2-Dimethylcyanoacetate Methyl Ester might not be a household name, its role in the world of chemistry is significant. Its intricate chemical structure and versatility make it a valuable component in various chemical processes and research endeavors. As scientists continue to explore its properties and applications, it adds to the ever-expanding knowledge base of organic chemistry, showcasing the enduring curiosity and innovation of the field.